According to the United States Pharmacopeia and the National Formulary (USP 40-NF 35*), analysis of alanine and aspartic acid, and their related compounds (organic acids) should be carried out with a column with L17 packing material and meets following requirements. The SUGAR SH1011 confirmed the requirements were met.
(Note) Alanine nor aspartic acid will not be detected in this method.
*The version at the time of the application acquisition.
System suitability requirements For aspartic acid:
Resolution (maleic acid and malic acid): ≥ 1.5
Relative standard deviation (RSD) for maleic acid, malic acid, and fumalic acid: ≤ 5.0%
For aspartic acid:
Resolution (maleic acid and malic acid): ≥ 1.5
Relative standard deviation (RSD) for maleic acid, malic acid, and fumalic acid: ≤ 10.0%
Sample: 10 µL
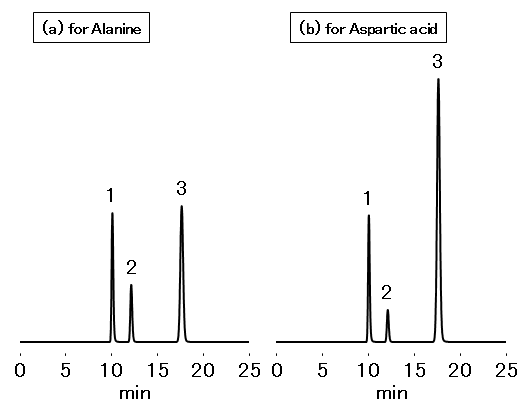
(a) Sysytem suitability solution for Alanine
- 1.Maleic acid 0.05 mg/mL
- 2.Malic acid 3 mg/mL
- 3.Fumaric acid 0.05 mg/mL
(b) Sysytem suitability solution for Asparatic acid
- 1.Maleic acid 0.05 mg/mL
- 2.Malic acid 1.5 mg/mL
- 3.Fumaric acid 0.1 mg/mLCa2+
- Column
- :Shodex SUGAR SH1011 (8.0 mm I.D. x 300 mm)
- Eluent
- :0.008 N H2SO4 aq.
- Flow rate
- :0.6 mL/min
- Detector
- :UV (214 nm)
- Column temp.
- :30 ℃
Sample Name Index
Operation Manual / Certificate of Analysis
Operation Manuals and Certificate of Analysis / Inspection Certificate for the following products can be downloaded here.
Product Name Index
Applications
- Effects of Flow Rate (KC-811)
- Effects of Injection Volume (KC-811)
- Effects of Sample Load (KC-811)
- Effects of Column Temperature (KC-811)
- Effects of Concentration of Eluent on Elution Volume (KC-811)
- Effects of Column Temperature on Elution Volume (1) (KC-811)
- Effects of Column Temperature on Elution Volume (2) (KC-811)
- Effects of Acetonitrile in Eluent (1) (KC-811)
- Effects of Acetonitrile in Eluent (2) (KC-811)
- Effects of Reversed Phase Column on Elution Volume (KC-811 + DE-613)
- Elution Volume of Aromatic Organic Acids (NN-814)
- Elution Volume of Organic Acids (NN-814)
- Elution Volume of Organic Acids (DE-413)
- Comparison of Analysis of Organic Acids with 4 Kinds of Detectors (Standards) (KC-811)
- Comparison of Analysis of Organic Acids with 4 Kinds of Detectors (Soy Sauce) (KC-811)
- Organic Acid Standards (1) (KC-811)
- Organic Acid Standards (2) (KC-811)
- Organic Acid Standards (3) (KC-811)
- Organic Acid Standards (4) (DE-413)
- Organic Acid Standards (5) (DE-213)
- Citric Acid in Phosphoric Acid (DE-613 + KC-811)
- Analysis of Organic Acids in Beer (KC-811)
- Analysis of Organic Acids and Vitamin C in Fruit Juice (KC-811)
- Reverse Phase Analysis of Gluconic Acids (DE-613)
- Analysis of Organic Acids in Sake (Japanese Rice Wine) (KC-811)
- Analysis of Organic Acids in White Wine (KC-811)
- Analysis of Oxalic Acid Using Ion Exclusion Mode (KC-811)
- Organic Acid in Phosphoric Acid (DE-613 + KC-811)
- Analysis of Organic Acids in Soy Sauces (KC-811)
- Organic Acids in Vinegar (KC-811)
- Separation of Succinic Acid and Fumaric Acid (KC-811)
- Analysis of Organic Acids (Eluent Conteinig Cyclodextrin) (KC-811)
- Camphor and Benzoic Acid (NN-814)
- Di-carboxylic Acids (DE-413)
- Analysis of Organic Acids in Soybean Paste (KC-811)
- Analysis of Organic Acids in Soy Sauce For Noodles (KC-811)
- Organic Acids Analysis in Black-vinegar (KC-811)
- LC/MS Analysis of Organic Acids (DE-213)
- Simultaneous Analysis of Saccharides, Organic Acids and Amino Acids with LC/MS (VG-50 2D)
- Simultaneous Analysis of Hydrophilic Substances in Energy Drink with LC/MS (VG-50 2D)
- Organic Acids (KC-811)
- LC/MS Analysis of Pipecolic Acid (NH2P-40 2D)
- Elution Volume of Organic Acids (NI-424)
- Analysis of Formic Acid in Povidone According to Pharmacopeia Method (KC-811)
- Analysis of Acetic Acid in Vinegar Drinks (KC-811)
- Citric Acid Analysis in Functional Beverage (KC-811)
- Organic Acid Determination Test for Glycerin Fatty Acid Esters According to Japan’s Specifications and Standards for Food Additives (SH1011) (SH1011)
