Chromium usually exists as trivalent chromium in nature and it is stable. On the other hand, hexavalent chromium has a very strong oxidative effect and is known to be very toxic. It may cause dermatitis and tumors when contacted and nasal mucosa damage when inhaled. It is also a carcinogen to be cautioned. Therefore, hexavalent chromium is a subject of regulation under many laws. In Japan, it is stated under Basic Environment Act/Environmental Quality Standards and Water Pollution Prevention Act/Effluent Standards. The "Measurement Manual for Hazardous Air Pollutants" prepared by the Ministry of the Environment in March 2018 includes a method for measuring hexavalent chromium in air dust. It uses an alkali-impregnated filter to collect the sample and uses ion chromatograph with post-column absorptiometry for the measurement: After the LC separation step, the analytes are derivatized using 1,5-diphenylcarbonohydrazide and detected by an ultraviolet-visible absorbance detector. The lower target quantification limit in this measurement is set at 0.08 ng/m3, because the 10-5 risk level standard stated by the United States Environmental Protection Agency (EPA) is 0.8 ng/m3, and is required to measure one tenth of it. If possible, it is even more desirable to be able to measure 0.025 ng/m3, which is one tenth of 0.25 ng/m3, the lower level standard in the WHO European Secretariat guideline.
In this application, hexavalent chromium standard reagent was measured using an IC SI-50 4E, an anion analysis column. Figure 1 shows chromatograms of blank and 0.1-, 0.5-, 1-, 2-, and 5-µg/L hexavalent chromium standard samples. The injection samples are extract of alkali-impregnated filter. And since these extracts are basic (pH 9 to 10), the standards are dissolved in a solvent containing 5-mM sodium carbonate and 5-mM sodium hydrogen carbonate. Figure 2 shows the calibration curve of hexavalent chromium. A good linear calibration (r=0.9999) was obtained for the concentration range of 0.1 to 5 µg/L.
Sample : 500 µL, K2Cr2O7 Std.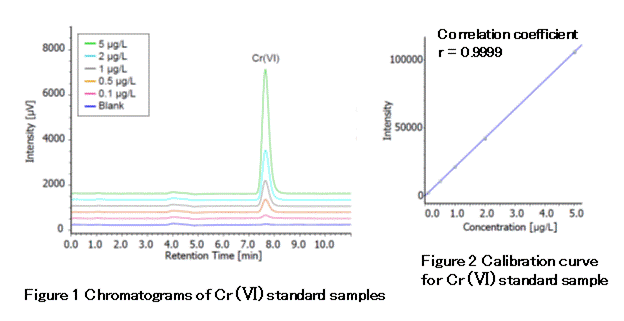
Figure 3 shows chromatograms of a 0.1-µg/L standard sample repeatedly injected (n=6). Table 1 summarizes the reproducibility of the measurement: CV% of the retention time and quantitative values were 0.14, and 1.58, respectively. The lower limit of quantification (S/N=10) calculated from the 0.1 µg/L chromatogram was 0.032 µg/L, and the lower limit of detection (S/N=3) was 0.009 µg/L. This lower limit of quantification corresponds to 0.022 ng/m3, when converted to air dust concentration*. Thus, this meets the WHO European Secretariat guideline target lower limit of quantification (0.025 ng/m3).
*Calculation for simulating a 5-mL volume, 5 L/min · 24 hr collection. Refer to Measurement Manual for Hazardous Air Pollutants.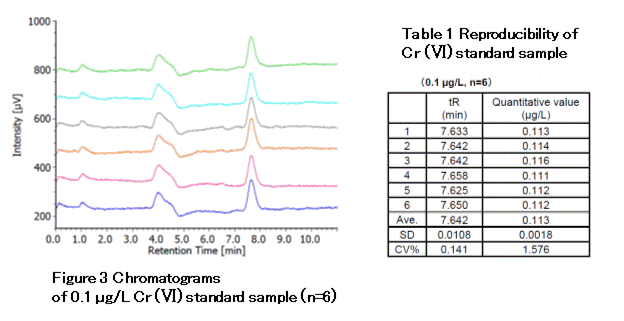
HPLC Data courtesy of JASCO Corporation
Please refer to the details on device configuration in Japanese.
Columns : Shodex IC SI-90G (4.6 mm I.D. x 10 mm)
+ SI-50 4E (4.0 mm I.D. x 250 mm)
Eluent : 40 mM Na2CO3 + 5 mM NaHCO3
Reagent : 2 mM 1,5-diphenylcarbonohydrazide-10 % methanol - 0.25 M sulfuric acid
Flow rate : (Eluent) 0.6 mL/min
(Reagent) 0.2 mL/min
Reaction temp. : 40 °C
Detector : VIS (540 nm)
Column temp. : 40 °C
Sample Name Index
Operation Manual / Certificate of Analysis
Operation Manuals and Certificate of Analysis / Inspection Certificate for the following products can be downloaded here.
