Cefepime is a type of cephems used to treat various infections. According to the United States Pharmacopeia and the National Formulary (USP 42-NF 37*), quantification of N-methylpyrrolidine, an organic impurity, in the cefepime injection should be carried out with a 4.0 mm (I.D.) x 25 cm (length) column with L76 packing material in 5 μm particle size and meets following requirements. The use of two IC YK-421 columns (12.5 cm each) in series confirmed the requirements were met.
System suitability requirements:
Relative standard deviation (RSD) of N-methylpyrrolidine peak area: ≤ 4.0%
*The version at the time of the application acquisition.
In addition to the method validation, a commercial cefepime dihydrochloride for injection was analyzed using the method. The concentration of N-methylpyrrolidine found was 0.09 %, confirming the detected amount was below the allowable limit (1.0 %). In this method, cefepime elutes around 50 minutes. It is detectable by a UV detector, but rarely detected by a CD detector. It is noteworthy that using one YK-421 column alone fulfills the system suitability requirements and it is applicable for the quantification of N-methylpyrrolidine in commercial cefepime injections.
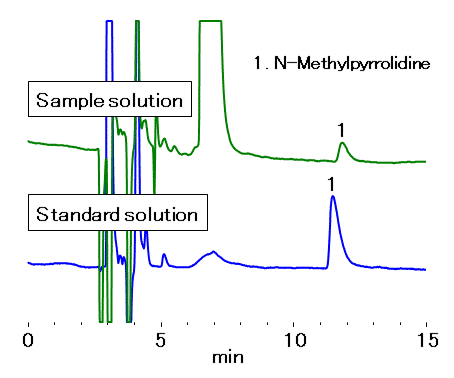
Sample : 10 μL each
(Sample solution) Commercial Cefepime hydrochloride formulation
Equivalent to 5 mg/mL of Cefepime hydrochloride in 0.002 N HNO3 aq.
(Standard solution) N-Methylpyrrolidine 0.05 mg/mL in 0.002 N HNO3 aq.
1. N-Methylpyrrolidine
Columns : Shodex IC YK-421 (4.6 mm I.D. x 125 mm) x 2 Eluent : 0.01 N HNO3 aq./CH3CN=19/1 Flow rate : 1.0 mL/min Detector : Non-suppressed conductivity Column temp. : Room temp.
Sample Name Index
Operation Manual / Certificate of Analysis
Operation Manuals and Certificate of Analysis / Inspection Certificate for the following products can be downloaded here.
Product Name Index
Applications
- Monovalent and Divalent Cation Standards (1) (YK-421)
- Monovalent and Divalent Cation Standards (2) (YK-421)
- Monovalent and Divalent Cation Standards (3) (Dipicolinic Acid + Crown Ether Eluent) (YK-421)
- Monovalent and Divalent Cation Standards (4) (YK-421) (Phosphoric Acid Eluent)
- Monovalent and Divalent Cation Standards (5) (YK-421) (Phosphoric Acid + 18-Crown 6-Ether Eluent)
- Monovalent and Divalent Cation Standards (6) (Comparison of YS-50 and YK-421)
- Monovalent and Divalent Cation Standards (7) (Eluent Conteinig Acetonitrile) (YS-50)
- Monovalent and Divalent Cation Standards (8) (Ethylenediamine Containing Sample) (YS-50)
- Dynamic Range of Column (YK-421)
- High Sensitive Detection of Cations (YK-421)
- Effects of Acetonitrile Concentration on Elution Volume (YK-421)
- Effects of Methanesulfonic Acid Concentration on Elution Time (YS-50)
- Effects of Flow Rate on Plate Number (YS-50)
- Effects of Temperature (YS-50)
- Effects of Sample Load on Plate Number (YS-50)
- Linearity of Calibration Curves (YS-50)
- Effects of Crown Ethers (YS-50)
- Tap Water (1) (YK-421)
- Analysis of Tap Water (2) (YK-412) (Phosphoric Acid Eluent)
- Monovalent and Divalent Cations Analysis in Tap Water (Ethylenediamine Containing Sample) (YS-50)
- Monovalent and Divalent Cations Analysis in River Water (Ethylenediamine Containing Sample) (YS-50)
- Mineral Water (YS-50)
- Sea Water (2) (YS-50)
- Red Wine (1) (YK-421)
- Red Wine (3) (YS-50)
- Serum (YK-421)
- Long Chain Alkylamines (YK-421)
- Trialkylamines (YK-421)
- Tetraalkylammoniums (YK-421)
- Analysis of Tetraalkylammoniums (YS-50)
- Alkylamines (2) (YK-421) (Phosphoric Acid + Acetonitrile)
- Ethanolamines and Alkylamines (YK-421) (Phosphoric Acid + Acetonitrile)
- Analysis of Ethanolamines and Alkylamines (YS-50)
- Cations and Alkylamines (YS-50)
- Analysis of Methylamines (YS-50)
- Analysis of Ethylamines (YS-50)
- Analysis of Short Chain Alkylamines (YS-50)
- Cation Analysis in the Presence of PEG (YS-50)
- Cation Analysis in the Presence of Albumin (YS-50)
