In the Guidelines on Nutrition Labelling in Japan, amino columns of 4.6 mm I.D. x 250 mm Length are adopted for the analysis of monosaccharides, disaccharides and sugar alcohols. This chromatogram shows the analysis result of typical sugar alcohol standards using an amino column, Asahipak NH2P-50 4E . Since the column is polymer-based, it is durable compared with silica-based columns and can be washed by alkali solution. The eluent used is the mixture of water and acetoritrile. The elution volume can be changed by changing the mixing ratio. 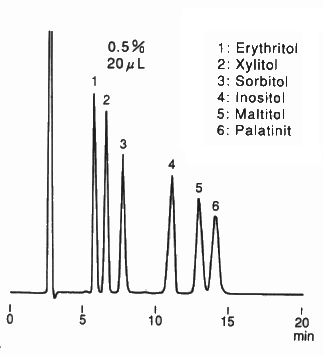
Sample : 0.5 % each, 20 μL
1. meso-Erythritol
2. Xylitol
3. Sorbitol
4. myo-Inositol
5. Maltitol
6. Palatinit
Column : Shodex Asahipak NH2P-50G 4A (4.6 mm I.D. x 10 mm)
+ NH2P-50 4E (4.6 mm I.D. x 250 mm)
Eluent : H2O/CH3CN=25/75
Flow rate : 1.0 mL/min
Detector : RI
Column temp. : Room temp.
Sample Name Index
Operation Manual / Certificate of Analysis
Operation Manuals and Certificate of Analysis / Inspection Certificate for the following products can be downloaded here.
Product Name Index
Applications
- Elution Volume of Saccharides and Sugar Alcohols (NH2P-50 4E)
- Sugar Alcohols (1) (SC1211)
- Sugar Alcohols (3)
- Saccharides and Sugar Alcohols (1) (SZ5532)
- Saccharides and Sugar Alcohols (2) (SP0810)
- Saccharides and Sugar Alcohols (3) (SP0810)
- Saccharides and Sugar Alcohols (4) (SC1011)
- Saccharides and Sugar Alcohols (5) (SZ5532)
- Saccharides and Sugar Alcohols (6) (NH2P-50)
- Saccharides and Amino Acids (1) (SC1011)
- Saccharides and Amino Acids (2) (SC1211)
- Saccharides and Amino Acids (3) (KS-801)
- Saccharides and Amino Sugars (2) (NH2P-50 4E)
- Saccharides and Amino Sugars (3) (SC1011)
- Xilitol in Tablet Candy
- Apple Juice (NH2P-50 4E)
- Apple Juice (SC1011)
- Sake (Japanese Rice Wine)
- Effects of Acetonitrile Concentration in Analysis of Sugar Alcohols
- Amino Sugars (SB-802.5 HQ)
- Soft Drink Containing Erythritol (NH2P-50 4E)
- Palatinit (SP0810, SC1011, SZ5532)
- Analysis of Pinitol (SP0810)
- Analysis of Meglumine (NN-814)
- LC/MS/MS Analysis of Meglumine (VC-50 2D)
- Analysis of Meglumine (YS-50)
- Separation of myo-Inositol and D-chiro-Inositol (SZ5532)
- Separation of Taurine and Inositol (NH2P-50 4E)
- Analysis of Hydrogenated Maltose Starch Syrup According to Japanese Pharmaceutical Excipients Method (KS-801)
- Analysis of Mannitol According to Pharmacopeia Method (SC1011-7F)
- Analysis of myo-Inositol According to EP Method (SC1011-7F)
- Analysis of Xylitol According to USP-NF Method (SP0810)
- Analysis of Sorbitol According to USP-NF Method (SP0810 8C)
- Analysis of Maltitol According to USP-NF Method (SP0810 8C)
- Comparison of Sensitivity of NH2P Polymer-based Column and Silica-based Amino Column with ELSD
- Analysis of Erythrose and Erythritol (VG-50 4E)
